In recent years, in addition to the growing demand for online purchases of regular products, there has been a rising consumer demand for online purchases of perishable goods such as seafood and meat. There are many foods, medicines and other commodities that need to be kept at a certain temperature when transported over long distances. This relies on cold chain logistics technology. This post will give you a detailed overview of the definition, components and solutions of cold chain logistics.
What is cold chain logistics?
Cold chain logistics refers to the transportation of temperature-sensitive food products under the right temperature environment at all times to ensure quality. Cold chain logistics is a logistics system that specializes in handling logistics that require temperature control. Generally such as ice cream, milk, meat, seafood, etc., need to be transported through cold chain logistics. Cold chain transportation mainly includes frozen transportation, refrigerated transportation and constant temperature transportation.
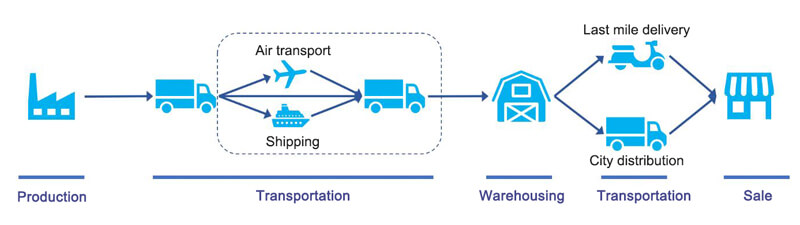
- Frozen transportation (-18℃~-22℃): Provide standard refrigerated transportation vehicles to transport goods such as frozen food, meat, ice cream and so on.
- Refrigerated transportation (0℃~7℃): Provide refrigerated transportation vehicles that meet the standards to transport goods such as fruits, vegetables, beverages, fresh milk products, flowers, plants and seedlings, cooked food products, cakes, various food materials and so on.
- Constant Temperature Transportation (18℃~22℃): Provide standard heat preservation and temperature control transportation vehicles to transport goods such as chocolates, candies, medicines, chemical products and so on.
The composition of cold chain logistics
1. Cold storage
Cold storage is the core of cold chain logistics. It controls temperature, humidity and air circulation through air conditioning or refrigerant circulation system to ensure that the goods remain fresh during storage.
2. Cold chain tools
Cold chain transportation tools are mainly divided into land transportation and sea transportation. Land transportation includes refrigerated trucks and refrigerated trains, etc., while sea transportation is based on refrigerated ships. Cold chain transportation needs to choose suitable transportation mode and tools according to the nature and demand of goods.
3. Temperature and humidity data loggers
Temperature and humidity data loggers can be used to record the temperature and humidity of the goods in the process of cold chain logistics. Choose suitable temperature recording equipment to track temperature changes during transportation, storage and distribution of goods. Temperature recording equipment generally includes temperature and humidity recorders and data collectors.

4. Packaging materials
Packaging materials are very important for cold chain logistics. Suitable packaging materials can prevent the goods from being affected by external temperature, humidity, pressure, vibration and other factors, so as to ensure the quality and safety of the goods during transportation.
5. Management platform
Management platform is a system used to monitor and manage the process of cold chain logistics, which is mainly used for real-time monitoring of information such as the location and temperature of goods, as well as providing data analysis and management services for enterprises. The construction of the data management platform needs to consider the stability of the system, data security and other aspects.
Challenges in cold chain logistics
- Inaccurate temperature monitoring. In the process of cold chain logistics, the temperature of each link needs to be strictly monitored. However, in the actual transportation process, temperature fluctuations will inevitably occur due to inaccurate thermometer readings or malfunctioning of the monitoring system.
- High transportation cost. Compared with ordinary logistics, the transportation cost of cold chain logistics is relatively high. Because cold chain logistics requires special storage, transportation and technology, the transportation cost will be relatively higher.
- Equipment failure: temperature control and refrigeration equipment are crucial for cold chain logistics. Failure of these systems during transportation may lead to temperature deviation and jeopardize the quality and safety of perishable goods.
- Influenced by weather. Cold chain logistics transportation has high requirements on the weather environment, which will be affected by the season, climate and other factors. If bad weather is encountered, it may bring losses to perishable or higher temperature products.
- High cost: Cold chain logistics usually requires expensive equipment and technology, which increases operating costs. Temperature monitoring, refrigeration equipment and maintenance of transportation vehicles all require significant capital.
- Energy consumption: Maintaining a low temperature environment requires energy consumption, a process that has a negative impact on the environment. The development of green cold chain technologies is becoming particularly important to reduce energy dependency.
- Compliance: Different countries and regions have different regulatory standards for cold chain logistics, which need to be adhered to when transporting across borders. Examples include Good Distribution Practice (GDP) and Hazard Analysis and Critical Control Points (HACCP).
Cold chain logistics solutions
For cold chain box, cold chain truck, cold chain warehouse, etc., the use of air temperature and humidity data logger, real-time display of temperature and humidity changes. Temperature and humidity data logger has both LCD screen real-time display data, but also supports real-time data upload for remote viewing. Once the temperature and humidity are out of range, the alarm will be raised immediately to remind the management personnel to check the working condition of the refrigeration equipment in time.
Cold chain monitoring involves front-end processing plants, logistics, distributors, stores and so on. The temperature range covers a wide range of scenarios involving chilling, freezing, ultra-low temperature and other specific monitoring solutions:
Cold storage room
Wired program with temperature and humidity sensor and management mainframe.
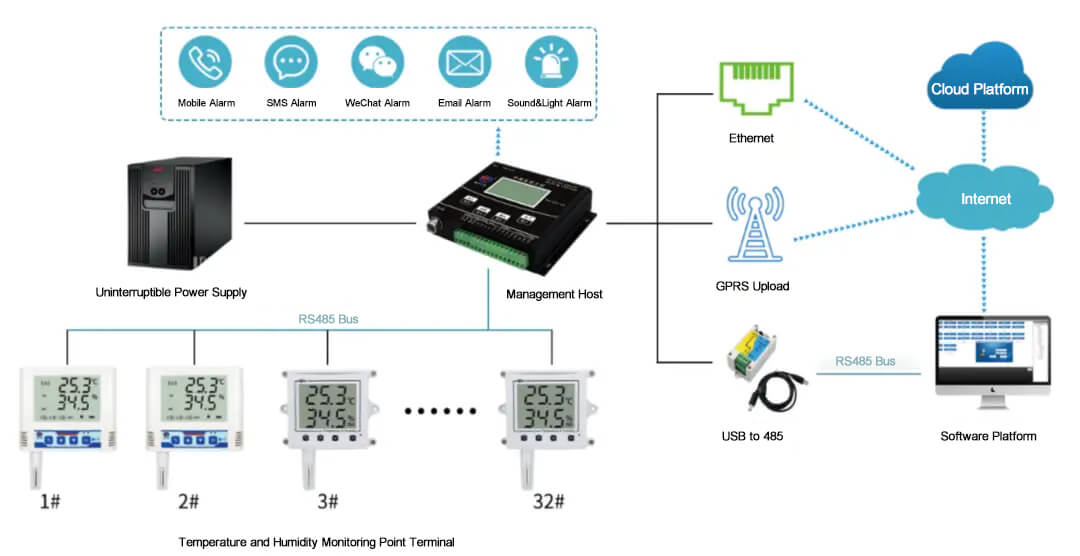
It is time-consuming and laborious to use thermometers to detect temperature and humidity in the warehouse. For this kind of location, it is suitable to adopt the solution of temperature and humidity sensor from the measurement point terminal with environment monitoring host, uninterruptible power supply and environment monitoring cloud platform. The collected data is uploaded to the cloud platform through the environment monitoring host in real time. The cloud platform has real-time monitoring, historical data query, multi-terminal view, abnormal data alarm and other functions. It also meets the requirements of GMP\GSP and other specifications to provide a good storage environment for cold storage.
Refrigerated truck
Wireless temperature and humidity data logger with wireless management host combination program.
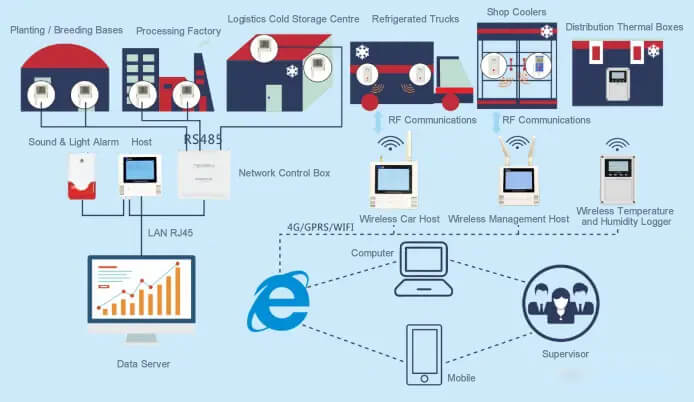
Refrigerated truck is a closed van-type transportation vehicle used to transport frozen or fresh goods, and it is a special transportation vehicle equipped with temperature and humidity monitoring equipment and refrigeration equipment. During cold chain transportation, goods need to be transported under certain temperature and humidity conditions to ensure quality and safety. Wireless temperature data logger can record the temperature and humidity data in the refrigerated compartment in real time, and transmit the data to the management platform through 4G/GPRS/WIFI/LORA and other wireless communication methods, so that the cold chain transportation managers can understand the transportation situation at any time.
Application of cold chain logistics
1. Fruit and vegetable cold chain
Fruit and vegetable cold chain refers to the fruit and vegetables in the picking, processing, transportation, storage of the whole process of the cold chain system, the respiratory system of fruits and vegetables to limit and reduce the volatilization of water, so as to effectively ensure that fruits and vegetables, nutrition and water sufficiency. The quality of fruits and vegetables and its moisture there is an inevitable relationship, moisture can reduce its nutritional cost of dissipation. Lower temperatures ensure adequate moisture. Usually, ordinary fruits say that the appropriate temperature is generally above and below zero degrees, but in practice need to make adjustments according to the different varieties.
2. Frozen food cold chain
For quick-frozen products, it is through certain technical means in the negative twenty-five degrees to let the product immediately frozen, in the transportation process need to maintain this lower temperature, so as to ensure that the product will not spoil. For example, the life of frozen dumplings, steamed buns and chaos. Frozen food on the storage and transportation requirements are very strict, must be guaranteed at -18 degrees Celsius below.
3. Aquatic cold chain
Aquatic cold chain is a way of cold chain treatment of aquatic products, covering all aspects of aquatic products from capture to storage, using this way not only to effectively ensure that the nutrients and freshness of seafood, but also to extend the storage period of seafood. No preservatives will be added in the whole process, which is an effective way to store aquatic products.
4. Meat cold chain
The processed meat is promptly placed in a low-temperature environment, so that the meat from the normal body temperature drops rapidly to about zero degrees, and then a day of acidification treatment, in the subsequent processing, distribution, storage and sales process are to ensure that the temperature is between zero degrees and minus forty degrees, thereby extending the storage time of meat products.
5. Medical Cold Chain
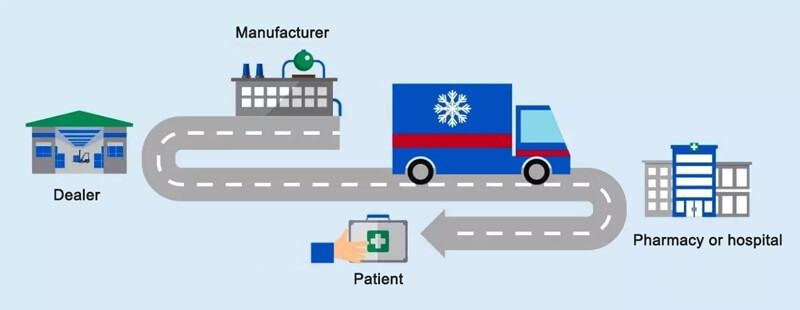
Some medical supplies (e.g. vaccines, blood products, etc.) are easy to change color, taste and mildew under room temperature conditions, and cannot keep quality. Only under low-temperature refrigeration conditions can the effectiveness be guaranteed, so the pharmaceutical cold chain transportation is thus generated. Compared with other cold chain systems, the requirements of pharmaceutical cold chain are more stringent. It not only requires relevant enterprises and practitioners to have high professional ability to ensure the safety of medicines; but also requires every relevant link to strictly comply with its temperature range to ensure the safety of patients’ lives.











