Biologists estimate that the human nose can distinguish on the order of ten thousand odor types, while dogs can identify hundreds of thousands to several million odor signatures and can isolate a specific odor from complex mixtures. Even so, a large number of gases in nature are colorless and odorless, including many that are toxic, asphyxiating, or explosive. These gases cannot be reliably detected by human or animal senses. For this reason, gas sensors are essential instruments for detecting gas presence and measuring gas concentration.
A gas sensor detects a specific gas and measures whether the gas is present near the sensor, or its content in the air. Therefore, gas sensors are often indispensable in safety systems. The gas sensor has many different types and can measure different gases. There are sensors that measure non-toxic gases such as oxygen and carbon dioxide, and sensors that detect toxic gases such as carbon monoxide, TVOC, and ammonia. Gas sensors are often used in flammable, explosive and toxic places to protect workers.
What is a gas sensor?
Gas sensors are devices that detect the presence of gases and measure their concentration by converting physical or chemical interactions into electrical signals. These signals are processed and expressed in standard units such as ppm, %LEL, or %Vol.
Gas sensors are widely used in industrial safety, environmental monitoring, medical systems, transportation, and residential applications. They are designed to monitor combustible gases, toxic gases, oxygen levels, and natural gas leaks in both open air and confined spaces.
From an engineering perspective, gas sensors are not just sensing elements. They are complete systems that integrate gas diffusion control, signal processing, environmental compensation, and long-term stability management to ensure reliable and accurate measurements.
Gas sensors types
There are many types of gas sensors. Selecting a suitable gas sensor requires understanding sensor characteristics, target gases, operating environment, and long-term stability requirements. Typical user concerns include: which sensor is selective to toxic gases, which sensor offers high precision, which sensor supports portable use, and which sensor is suitable for continuous fixed monitoring.
- According to different gas types, sensors can be divided into combustible gas sensors (often catalytic combustion, infrared, thermal conductivity, semiconductor), toxic gas sensors (often electrochemical, metal oxide semiconductor, photoionization), oxygen sensors (paramagnetic, zirconia), and special industrial sensors.
- According to usage methods, they can be divided into portable detectors and fixed online detectors.
- According to sampling methods, they can be divided into diffusion sensors and pumped sensors. Diffusion sensors rely on natural gas diffusion to the sensing element. Pumped sensors actively draw gas into a measurement chamber, enabling faster response and remote sampling.
- According to functional integration, they can be divided into single gas sensors and multi gas composite sensors.
- According to detection principles, gas sensors can be classified into semiconductor, electrochemical, infrared (NDIR), catalytic, thermal conductivity, magnetic, photoionization (PID), acoustic/photoacoustic, mass-sensitive (SAW/QCM), colorimetric, and other specialized sensing technologies.
Classification by working principle is the most widely adopted approach. The following article will introduce you to eight gas sensors in detail, as well as their principles, advantages, limitations, and typical applications:
1. Semiconductor gas sensors
A semiconductor gas sensor, often based on metal oxide materials such as SnO₂ or ZnO, measures gas concentration through changes in electrical resistance caused by surface chemical reactions. At elevated operating temperatures, oxygen molecules adsorb on the sensor surface and capture free electrons, increasing resistance. When a reducing gas such as methane or hydrogen is present, it reacts with adsorbed oxygen, releasing electrons back into the conduction band and decreasing resistance.
The magnitude of resistance change correlates with gas concentration and is converted into an electrical output through a signal processing circuit.
Advantages:
Simple structure, low manufacturing cost, high sensitivity to many combustible and reducing gases, fast response.
Disadvantages:
Limited selectivity, strong cross sensitivity to other gases, significant influence from temperature and humidity.
Semiconductor sensors are widely used in domestic gas leak alarms and industrial combustible gas detectors for methane, LPG, hydrogen, and alcohol vapors.
2. Electrochemical gas sensors
Electrochemical sensors react with the target gas and generate an electrical signal proportional to gas concentration. Most electrochemical gas sensors are amperometric devices that output a current linearly related to the gas concentration.
The working principle of electrochemical gas sensors is as follows: the gas first passes through a diaphragm or porous membrane, which prevents condensation and dust from entering the sensor. Gas molecules then diffuse through the filter layer and reach the working electrode, where oxidation or reduction reactions occur. These electrochemical reactions cause a change in the current flowing through the external circuit, which is proportional to the gas concentration.
Sensors based on electrochemical working principles adopt a three-electrode configuration, consisting of a working electrode, reference electrode, and counter electrode, to improve stability and linearity. Sometimes also use simpler two-electrode structures.

Advantages:
High sensitivity, excellent linearity, low power consumption, and good selectivity for specific toxic gases.
Disadvantages:
It needs to be detected in an environment with oxygen (1-10% VOL), and liquid electrolytes need to be consumed during use. Easily disturbed by temperature, humidity, pressure, and gases with similar chemical properties.
Electrochemical sensors are widely used for carbon monoxide, hydrogen sulfide, sulfur dioxide, nitrogen dioxide, ammonia, oxygen, and ozone detection in portable and fixed monitoring instruments.
3. NDIR gas sensors
NDIR(non-dispersive infrared) gas sensors measure gas concentration using infrared absorption. Gas molecules absorb specific infrared wavelengths corresponding to their molecular vibration modes. An infrared source emits broadband radiation through a gas chamber. The target gas absorbs characteristic wavelengths, and a detector measures the reduction in transmitted intensity. Using optical filters and reference channels, the sensor converts absorption into gas concentration.
Gas molecules vibrate at different frequencies. Some atoms are very small and light, so they vibrate at high frequencies. For example, the methane molecule’s vibration frequency is 333KHz, and the central wavelength is 3.3um. Because the molecular weight is different, the infrared absorption wavelength is also different, as shown in the following figure:
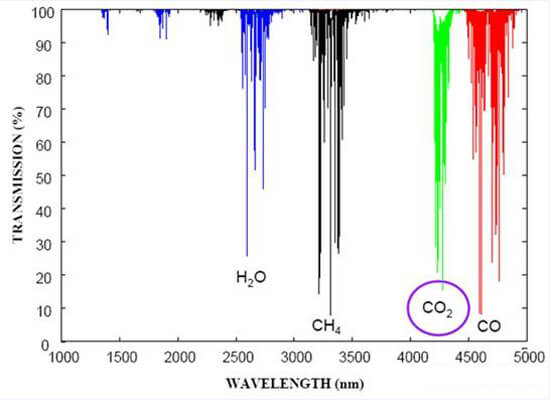
Advantages:
1. Common sensors on the market, such as catalytic combustion (CAT), electrochemical (EC), and semiconductor sensors, cannot measure any gas. For example, CO2 cannot be measured, but NDIR sensors can measure CO2. The most commonly measured gases for NDIR are CH4 and CO2.
2. Oxygen is not required. The CAT sensor requires oxygen to participate in the oxidation reaction. But NDIR is an optical sensor and doesn’t require oxygen to be involved.
3. The measurement concentration can reach 100%v/v. Because the NDIR sensor signal characteristics are: in a normal environment, the signal strength is the largest; the greater the measured gas concentration, the smaller the signal.
4. Good stability. NDIR sensor stability basically depends on the light source. As long as a good light source is selected, the long-term stability will be excellent, and it can be achieved without calibration for 2 years.
5. Low maintenance cost.
Disadvantages:
Higher power consumption, more complex optical and electronic structure, higher cost, and relatively higher expense for ppm-level detection.
NDIR sensors are the primary solution for carbon dioxide, methane, and refrigerant gas monitoring in environmental, industrial, and safety systems.
4. Catalytic gas sensors
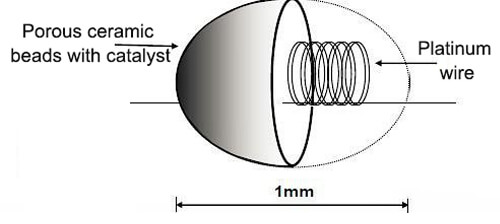
This sensor detects combustible gases by catalytic oxidation. A platinum coil embedded in a porous ceramic bead is coated with catalyst. When combustible gas contacts the bead at elevated temperature, oxidation occurs, releasing heat and increasing the bead’s resistance. The resistance change is proportional to combustible gas concentration and is measured using a Wheatstone bridge circuit.
Since the catalytic gas sensor platinum resistance is usually wrapped by porous ceramic beads, it is also called a catalytic bead gas sensor. In theory, this sensor can detect all combustible gases, but there are many exceptions in practical applications. This sensor can usually be used to detect combustible gases such as methane, LPG, acetone, etc. in the air.
Advantages:
Strong resistance to harsh climate and poisonous gas, long service life. Can detect all flammable gases, including alkanes and non-alkanes. Low maintenance cost.
Disadvantages:
Work in the dark, easy to explode or catch fire. Components are susceptible to poisoning by sulfide and halogen compounds, which shorten their service life. In a hypoxic environment, the error is larger.
This sensor is mainly used to detect combustible gases, such as gas generating stations, and gas plants to analyze CO, H2, C2H2, and other combustible gases in the air.
5. Magnetic gas sensors
After the magnetic gas sensor senses heat, light, radiation and pressure in the environment, its magnetic properties will also change accordingly. Using this feature, various reliable and high-sensitivity sensors can be made. Magnetic gas detectors are mostly magnetic probes with relatively strong measurement capabilities. When measuring gases, it is primarily used for oxygen measurement. Oxygen is strongly paramagnetic, while most other gases are weakly diamagnetic. Two common types are magneto-mechanical sensors and thermomagnetic convection sensors.
Advantages:
Excellent selectivity for oxygen, high precision, long-term stability, and no chemical consumption.
Disadvantages:
Limited to oxygen measurement, higher cost, and relatively complex mechanical structures.
It is commonly used in chemical fertilizer production, cryogenic air separation, thermal power station combustion system, natural gas to acetylene, and other industrial production oxygen measurement. Monitor exhaust gas, tail gas, flue gas and other emissions.
6. Photoionization gas sensors
Photoionization sensors (PID sensors) work by using an ultraviolet lamp to ionize gas molecules whose ionization energy is lower than the lamp energy. The generated ions and electrons move between electrodes under an electric field, producing a current proportional to the gas concentration. This principle makes PID sensors especially suitable for detecting volatile organic compounds and many toxic industrial gases.
Advantages:
PID sensors offer very high sensitivity, fast response, and low power consumption because they operate at ambient temperature. They can detect a wide range of VOCs at ppm to ppb levels and are well suited for portable instruments.
Disadvantages:
They are non-selective, cannot detect permanent gases such as methane, carbon monoxide, or carbon dioxide, and their performance can be affected by humidity and lamp contamination, requiring periodic maintenance.
PID sensors are widely used in industrial hygiene, environmental monitoring, leak detection, and emergency response equipment. Typical applications include workplace exposure monitoring, petrochemical facilities, indoor air quality assessment, soil gas surveys, and hazardous material detection.
7. Thermal conductivity gas sensors
A thermal conductivity gas sensor is a device that can sense a certain gas in the environment. It converts gas concentration information into electrical signals for detection, monitoring, analysis and alarming. Thermally conductive gas-sensitive materials measure their concentration based on the difference in thermal conductivity between different gases and air. Usually, a change in thermal conductivity is translated into a change in resistance through the circuit. The gas type and level are calculated from the change in resistance value.
Advantages:
Wide detection range, the highest detection concentration can reach 100%. Good working stability, long service life, no catalyst aging problem.
Disadvantages:
Low sensitivity, limited selectivity, strong temperature dependence, and poor performance at trace concentration levels.
They are commonly used for hydrogen, helium, and binary gas mixtures in industrial process monitoring.
8. Gas chromatograph analyzer
Based on chromatographic separation technology and detection technology, various gas samples are separated and measured. Hence the full analytical sensor. It has been used in power plant boiler tests.
During work, a certain gas sample is periodically taken from the sampling device. The chromatographic column is carried by a pure carrier gas (ie mobile phase) with a certain flow rate, and the chromatographic column is filled with a solid or liquid called a stationary phase. The components are repeatedly distributed in the two phases, and flow out of the chromatographic column according to time and enter the detector for quantitative determination.
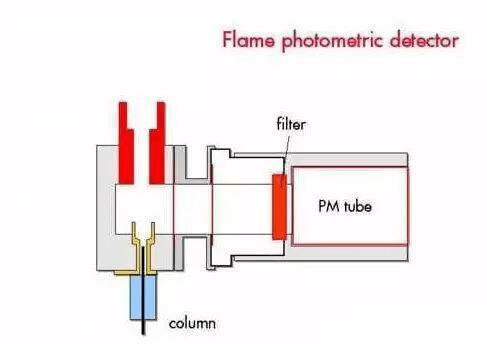
Advantages:
High sensitivity, suitable for micro and trace analysis, and can analyze complex multiphase separation gases.
Disadvantages:
Regular sampling cannot achieve continuous sampling and analysis, and the system is more complicated. It is mostly used for laboratory analysis, and is not suitable for industrial field gas monitoring.
Gas sensors are essential devices for detecting and quantifying gaseous substances in both industrial and civil environments. Each type relies on a distinct physical or chemical mechanism, resulting in different performance characteristics in terms of sensitivity, selectivity, response time, stability, and operating conditions. From a development perspective, a reliable gas sensor is not merely a sensitive element, but an integrated system that combines gas diffusion control, signal conditioning, environmental compensation, and long-term stability management. Understanding the working principles, advantages, and limitations of different gas sensor technologies is essential for selecting appropriate solutions for safety monitoring, environmental measurement, and process control applications.

This article was written by the Renke Technical Team. The team focuses on collecting, analyzing, and addressing real customer challenges, and shares practical solutions through technical articles to help other users solve similar problems. Renke is a trusted sensor manufacturer with more than 15 years of hands on experience in hardware and software research and development.